Exploring Hf Lewis Structure: Key Concepts & Examples
by Ferrara
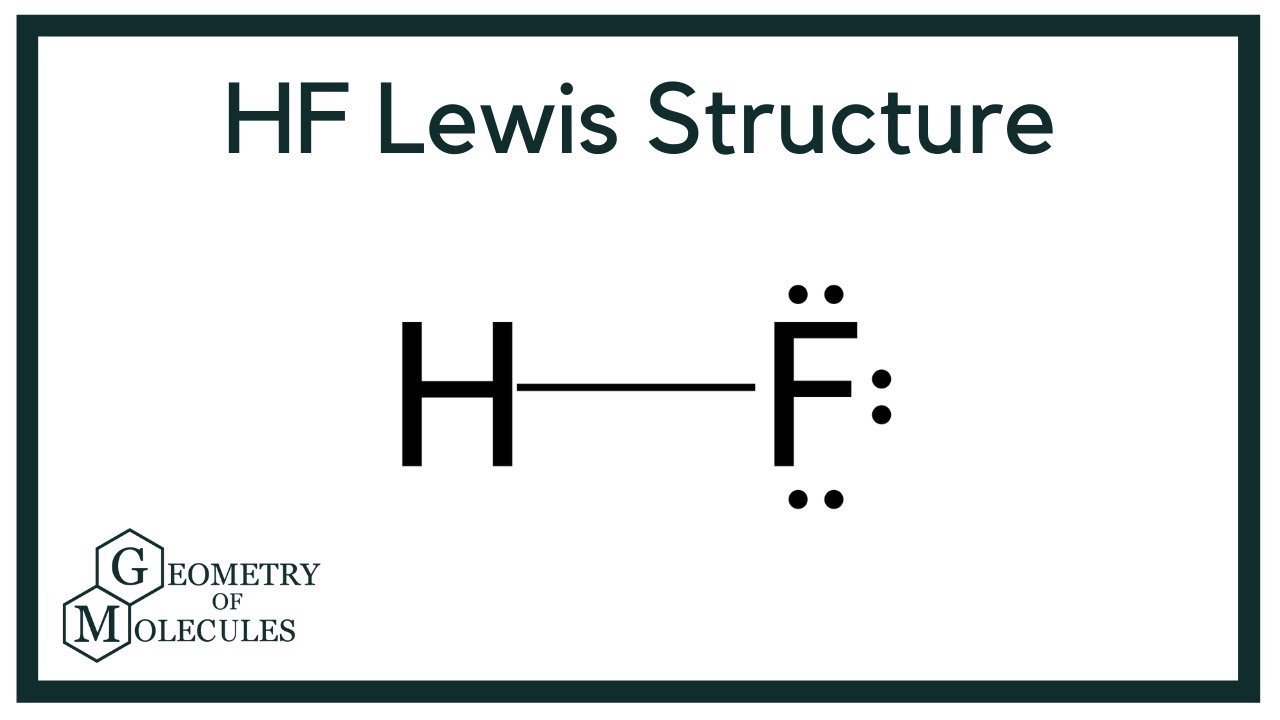
The HF Lewis structure? It’s all about getting the right arrangement of atoms and electrons. Picture this: hydrogen bonded to fluorine, creating a dynamic duo in the world of chemistry. Electrons are shared, forming a stable bond. Understanding the HF Lewis structure is key to unraveling the mysteries of chemical bonding. Join me as we delve into the fascinating realm of molecular geometry and discover the beauty of HF’s simple yet intricate structure.
The H F Lewis Structure: A Guide to Understanding Chemical Bonding
Welcome, young scientists! Today, we are going to delve into the fascinating world of chemistry to explore the HF Lewis Structure. Have you ever wondered how atoms come together to form molecules? Well, the Lewis Structure is like a roadmap that helps us understand how atoms bond and share electrons to create stable compounds. So, let’s put on our lab coats and goggles as we embark on this exciting journey!
What is the HF Molecule?
Before we dive into the Lewis Structure of HF, let’s first understand what HF actually is. HF stands for Hydrogen Fluoride, a chemical compound composed of hydrogen and fluorine atoms. Hydrogen Fluoride is a colorless gas with a sharp odor and is commonly used in industrial processes and as a refrigerant.
Understanding Chemical Bonding
But how do these hydrogen and fluorine atoms bond together to form HF? Well, it all comes down to the concept of chemical bonding. Atoms are like tiny puzzle pieces that want to connect with each other to become stable. In the case of HF, hydrogen and fluorine atoms are attracted to each other due to their opposite charges.
The Role of Electrons
Electrons play a crucial role in chemical bonding. They are like the glue that holds atoms together. In the HF molecule, hydrogen has one electron, while fluorine has seven electrons in its outer shell. Fluorine wants one more electron to complete its outer shell and become stable.
So, what happens when hydrogen and fluorine come together? Hydrogen is willing to share its electron with fluorine to help it achieve stability. This sharing of electrons between atoms is what we call a covalent bond. In the case of HF, the shared electron pair forms the bond between hydrogen and fluorine.
The Lewis Structure of HF
Now, let’s take a closer look at the Lewis Structure of HF. The Lewis Structure is a simple way to represent how atoms are bonded in a molecule. In the case of HF, we have one hydrogen atom and one fluorine atom coming together to form a molecule.
In the Lewis Structure of HF, we draw the hydrogen atom with its single electron and the fluorine atom with its seven electrons. The shared electron pair between hydrogen and fluorine represents the covalent bond between the two atoms.
Drawing the Lewis Structure
To draw the Lewis Structure of HF, we start by placing the hydrogen atom on one side and the fluorine atom on the other side. We then connect the two atoms with a single line to represent the shared pair of electrons. Remember, each atom wants to achieve stability by having a full outer shell of electrons.
Hydrogen is happy because it can achieve stability with just two electrons in its outer shell. On the other hand, fluorine now has a total of 8 electrons in its outer shell, making it stable as well. This sharing of electrons allows both hydrogen and fluorine to achieve a stable configuration, just like completing a puzzle!
Properties of HF
Now that we understand the Lewis Structure of HF, let’s explore some of the properties of this fascinating molecule. HF has unique characteristics that make it useful in various applications.
Acidic Nature
Hydrogen Fluoride is considered a weak acid. When dissolved in water, HF can release hydrogen ions, making the solution acidic. This acidic nature of HF is why it is commonly used in industrial processes, such as etching and glass cleaning.
Reactivity
Due to the polar nature of the HF molecule, it is highly reactive. This reactivity makes HF useful in chemical reactions, such as in the production of fluorine-containing compounds. However, it also requires careful handling due to its corrosive properties.
Boiling Point and Melting Point
HF has a low boiling point and melting point compared to other hydrogen halides. This property makes it a valuable compound for various applications, including as a refrigerant and in the manufacturing of pharmaceuticals.
Congratulations, young scientists! You have successfully learned about the HF Lewis Structure and gained insight into the world of chemical bonding. Remember, the Lewis Structure is like a secret code that helps us unlock the mysteries of how atoms come together to form molecules.
Keep exploring the wonders of chemistry, and who knows what other exciting discoveries you might make in the world of science! Stay curious, stay inspired, and always keep your goggles on!
Draw the Lewis Structure of HF (hydrogen fluoride)
Frequently Asked Questions
What is a Lewis structure for HF molecule?
A Lewis structure for the HF molecule shows the arrangement of atoms, valence electrons, and bonds within the molecule. In the case of HF, hydrogen (H) forms a single bond with fluorine (F), and there are two lone pairs of electrons on the fluorine atom.
How many valence electrons are there in the HF Lewis structure?
In the HF Lewis structure, hydrogen contributes 1 valence electron while fluorine contributes 7 valence electrons, totaling 8 valence electrons in the molecule.
What is the molecular geometry of HF based on its Lewis structure?
Based on the Lewis structure of HF, the molecule has a linear geometry. This means that the hydrogen and fluorine atoms lie in a straight line with a bond angle of 180 degrees.
Final Thoughts
In conclusion, understanding the HF Lewis structure is crucial for grasping the chemical bonding in the HF molecule. The sharing of electrons between hydrogen and fluorine creates a polar covalent bond, with fluorine being more electronegative. This unequal sharing leads to a partial negative charge on fluorine and a partial positive charge on hydrogen. By knowing the HF Lewis structure, one can predict the molecule’s properties and behavior accurately. Mastering this fundamental concept enhances one’s understanding of chemical structures and interactions.
The HF Lewis structure? It’s all about getting the right arrangement of atoms and electrons. Picture this: hydrogen bonded to fluorine, creating a dynamic duo in the world of chemistry. Electrons are shared, forming a stable bond. Understanding the HF Lewis structure is key to unraveling the mysteries of chemical bonding. Join me as we…
Contact Us
[contact-form-7 id=”142″ title=”Contact form 1″]
Archives
- March 2026
- February 2026
- January 2026
- November 2025
- October 2025
- September 2025
- August 2025
- July 2025
- June 2025
- May 2025
- April 2025
- March 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- May 2024
- March 2024
- February 2024
- January 2024
- December 2023
- October 2023
- September 2023
- August 2023
- July 2023
- June 2023
- May 2023
- April 2023
- March 2023
- January 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- February 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- August 2020
- June 2020
- May 2020
- March 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- August 2019
- July 2019
- June 2019
- May 2019
- April 2019
- March 2019

Categories
- Loft Ladder
- Adventure
- asbestos
- Automotive
- Banking and company services
- Branding
- Business
- CANADA
- casino
- Country
- crypto
- Dating
- Entrepreneur
- Featured
- Finance
- gaming
- health
- Home Improvement
- INDIA
- Industry
- Internet marketing
- Law
- loans
- Marketing
- Merchant
- News
- Online Gaming
- Philippines
- real estate
- SEO
- Shopping
- sports
- Streaming
- Trading
- Transactions
- UK
- USA
- weed
Recent Posts
- Zrozumienie szans w buchmacherskich zakładach sportowych
- Акции в онлайн казино Пинко: последние новости
- Обзор Бк Пин Ап: надежный сервис для онлайн игры без риска
- Как быстро зарегистрироваться на Pin Up сайт с регистрацией и входом
- Обзор провайдеров игровых автоматов в 1хБет для игроков
- Обратите внимание: скачать 1хбет на айфон безопасно?
- Игровые стратегии для 1хбет мобильное приложение: советы экспертов
- Ценности выше всего: какие игры есть в казино Мостбет?
- На Пин-ап: предпочитаете классику? Игры со слотов для вас!
- Бессмысленные ставки: необычные рынки на 1xbet

